Graphite Fines: A Comprehensive Guide to Properties and Applications
Graphite fines, a versatile form of carbon, are gaining prominence across diverse industries due to their unique properties. From metallurgical applications to advanced material development, understanding the characteristics and uses of graphite fines is crucial for professionals and researchers alike. This article provides a detailed exploration of graphite fines, covering their properties, manufacturing processes, applications, and considerations for selecting the right grade for your specific needs. We'll delve into how Dah Carbon provides high-quality graphite fines for various industrial purposes.

What Are Graphite Fines?
Graphite fines refer to finely divided particles of graphite, typically with a particle size ranging from a few micrometers to several millimeters. They are produced through various processes, including milling and micronization of natural or synthetic graphite. Graphite itself is an allotrope of carbon, known for its exceptional thermal and electrical conductivity, high lubricity, and chemical inertness. These properties are retained and often enhanced in the fine particle form, making graphite fines invaluable in numerous applications. The purity and crystalline structure significantly impact the performance characteristics of these fines.
Key Characteristics: High electrical conductivity, excellent thermal conductivity, strong lubricity, chemical inertness, and high purity.
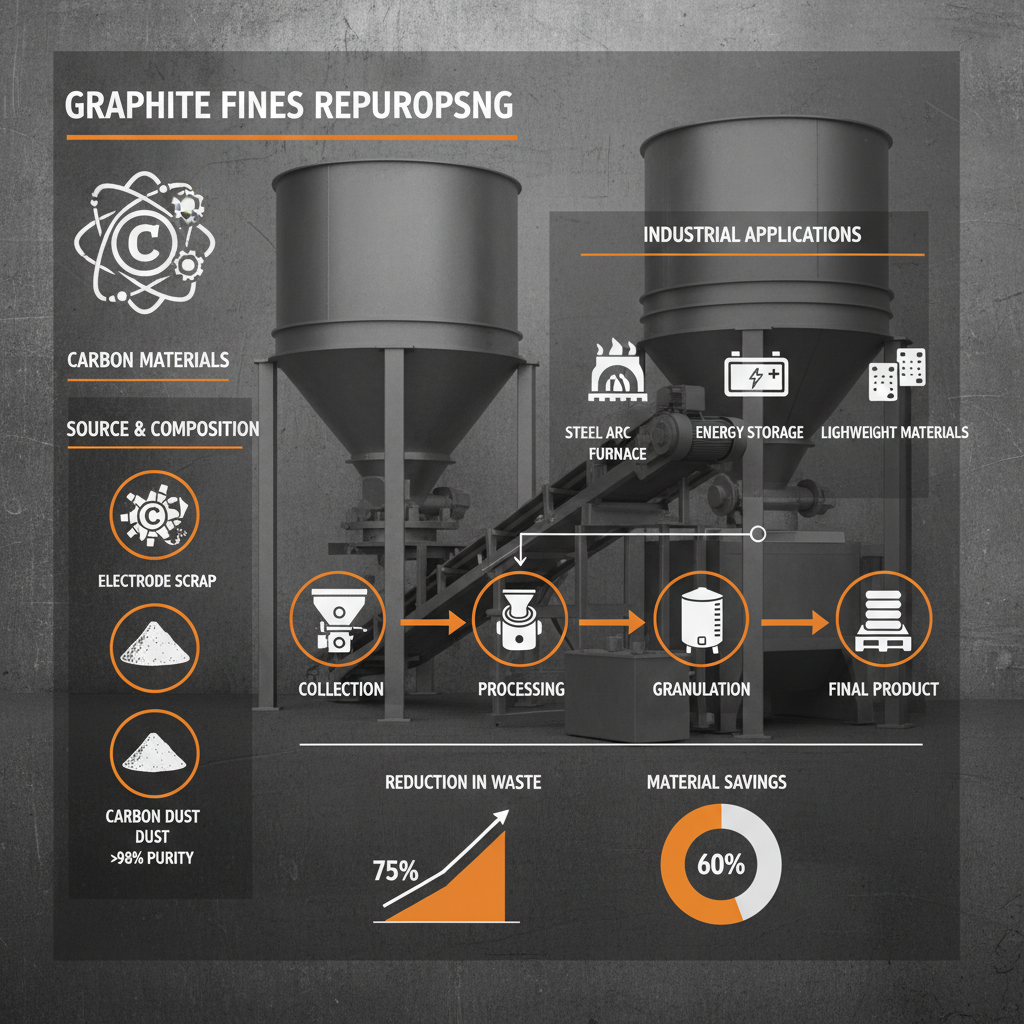
Manufacturing Processes for Graphite Fines
The production of graphite fines involves several techniques. Natural graphite fines are obtained by crushing, grinding, and classifying natural graphite ore. Synthetic graphite fines are created through the high-temperature treatment of carbonaceous materials like petroleum coke or coal tar pitch. Milling and micronization are crucial processes used to achieve the desired particle size distribution. Jet milling and ball milling are common methods. The choice of manufacturing process impacts the cost, purity, and crystalline structure of the final product. Dah Carbon employs advanced milling technologies to produce graphite fines with consistent particle size and high purity.
Key Applications of Graphite Fines
Graphite fines find applications across a wide spectrum of industries. In metallurgy, they are used as a carbon raiser in steelmaking and as a component in brake linings. They are essential in the production of lubricants, paints, plastics, and powder metallurgy. The increasing demand for lithium-ion batteries has driven a significant surge in the use of graphite fines as an anode material. Their unique properties enhance battery performance, including capacity and cycle life. Further, they are utilized in the manufacturing of friction materials, conductive polymers, and various specialized coatings.
Major Industries Utilizing Graphite Fines:
• Metallurgy
• Battery Manufacturing
• Lubricants
• Plastics & Polymers
• Coatings
Graphite Fines: Grades and Specifications
Graphite fines are available in various grades, differentiated by particle size, purity, and crystalline structure. Selecting the appropriate grade is critical for optimizing performance in specific applications. Finer particle sizes generally offer higher surface area and improved dispersion, while higher purity levels are essential for sensitive applications like battery manufacturing. Below is a table outlining typical specifications for different grades:
Selecting the Right Graphite Fine Supplier
Choosing a reliable supplier is paramount to ensure consistent quality and timely delivery. Consider factors such as manufacturing capabilities, quality control procedures, technical support, and commitment to sustainability. Dah Carbon offers a comprehensive range of graphite fines, backed by rigorous quality testing and dedicated customer support. We prioritize sustainable practices and are committed to delivering products that meet the highest industry standards.
Conclusion
Graphite fines are essential materials with applications spanning multiple industries. Understanding their properties, manufacturing processes, and grades is crucial for optimal performance. Dah Carbon is a trusted supplier, providing high-quality graphite fines and dedicated customer support.
Frequently Asked Questions (FAQs)
What factors influence the cost of graphite fines?
Several factors determine the price of graphite fines, including the type of graphite (natural vs. synthetic), particle size, purity, and order volume. Synthetic graphite fines generally command a higher price due to the more complex manufacturing process. Finer particle sizes and higher purity levels also contribute to increased costs. Market demand and transportation expenses can also impact pricing. Contacting Dah Carbon directly will provide you with a customized quote based on your specific requirements.
Are graphite fines hazardous to handle?
While graphite fines are generally considered non-toxic, inhalation of high concentrations of dust should be avoided. Prolonged exposure can cause respiratory irritation. It’s recommended to wear appropriate personal protective equipment (PPE), such as a dust mask and gloves, when handling graphite fines. Ensure adequate ventilation in the work area. Refer to the Material Safety Data Sheet (MSDS) for detailed safety information.
What is the shelf life of graphite fines?
Graphite fines have a virtually unlimited shelf life when stored properly. They are chemically inert and do not degrade over time. However, it’s essential to protect them from moisture and contamination. Store in a dry, sealed container in a well-ventilated area. Avoid exposure to direct sunlight and extreme temperatures.
Can graphite fines be used in conductive inks?
Yes, graphite fines are widely used in the formulation of conductive inks. Their excellent electrical conductivity and fine particle size make them ideal for creating printable conductive traces. These inks are employed in various applications, including printed electronics, sensors, and touchscreens. The specific grade of graphite fines and the ink formulation determine the conductivity and printability characteristics.


















